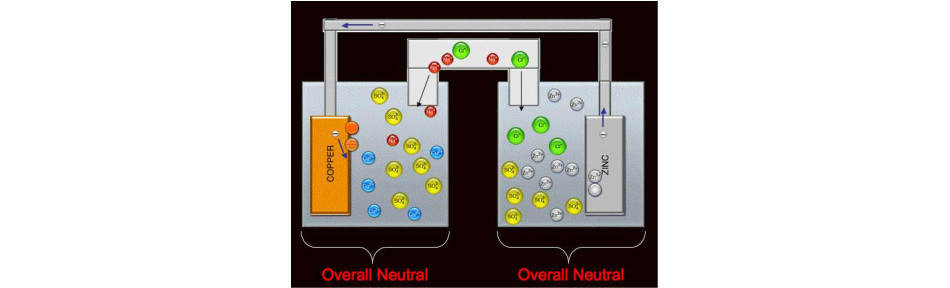
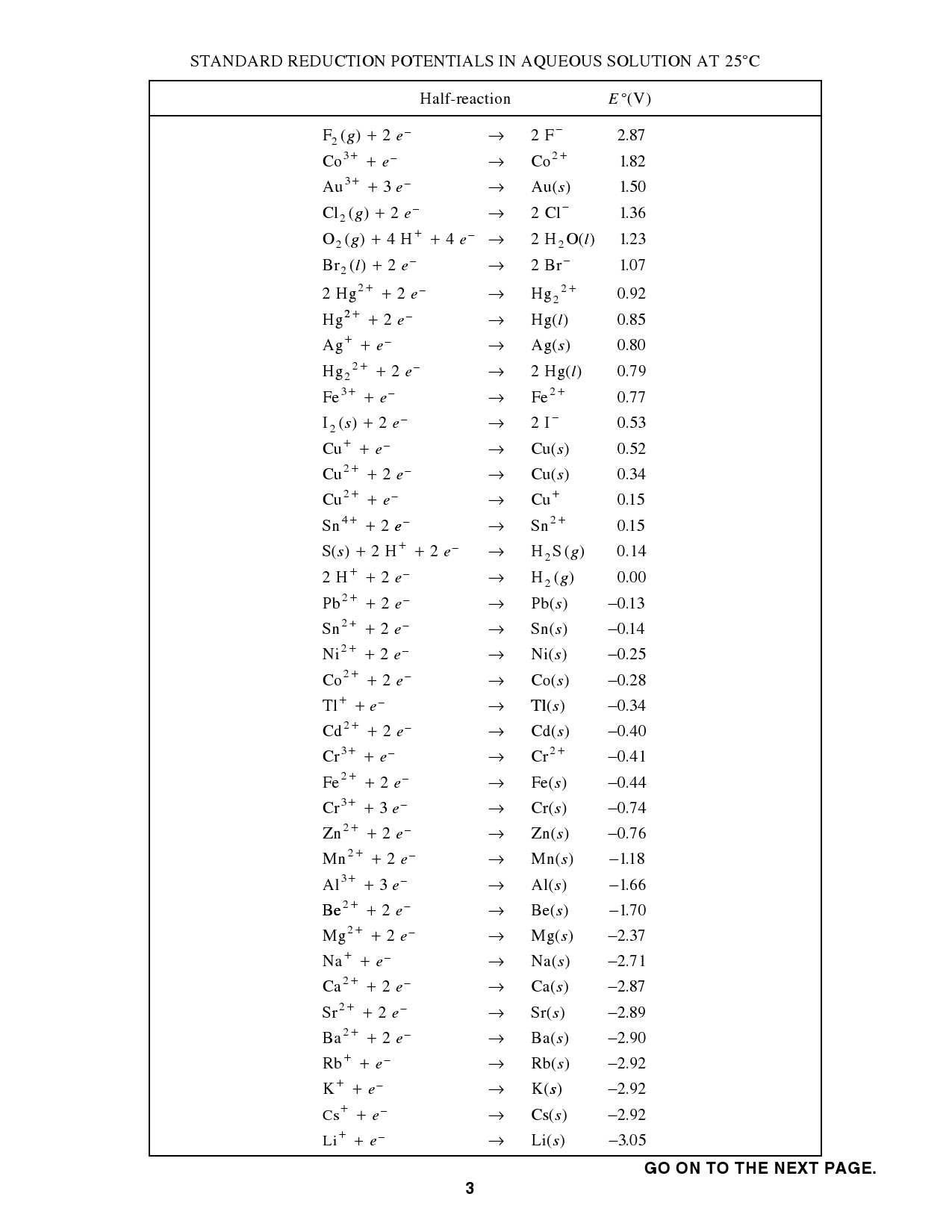
Using the standard reduction potentials listed in Table 20.1 or your AP Chem packet, calculate the standard emf for the voltaic cell described in Sample Exercise ...
17.2 Standard Reduction Potentials ... -Standard Reduction Potential, or E0, is a measure of the potential of a half-reaction to reduce at standard conditions (1M and .... The sign is reversed as well. The oxidation potential for Zn is +0.76V. Page 39. AP Chemistry.. Source of data: CRC Handbook of Chemistry and Physics, 84th Edition (2004). Previous Section · Table of Contents · Next Section.. There once was a table of reduction potentials in the reference tables of the AP Chemistry exam. Currently, we expect the data will be given in either a small .... ... Settings\Owner\Desktop\Website\apsrpotentials.doc. Page 1 of 1. STANDARD REDUCTION POTENTIALS IN AQUEOUS SOLUTION AT 25oC. Half-reaction.
ap chemistry reduction potential table
ap chemistry reduction potential table, chemistry standard reduction potential table, chemistry reduction potential table, reduction potential table ap chemistry Archicad 13 Francais Gratuit Avec Crackl
STANDARD REDUCTION POTENTIALS IN AQUEOUS SOLUTION AT 25 C. ∞. Half-reaction. (V). E∞. 2. F ( ) 2 g e. -. +. Æ. 2F. -. 2.87. 3+. Co e. -. +. Æ. 2. Co. +.. The Standard Reduction Potential Chart for chemistry classrooms lists 30 of the most useful reduction half-cell reactions are listed in descending order of .... Mar 6, 2021 — This can be done using an activity series. The table shown below is simply a table of standard reduction potentials in decreasing order. The .... Mar 11, 2015 — Identify the oxidizing and reducing agents. Solution. Using Table 17.2, the reactions involved in the galvanic cell, both written as reductions, are.. it the stronger oxidizing agent. 6) Use the list of standard reduction potential values to indicate which species in each set is the strongest reducing agent. a ... Perfectly Clear Complete 3.11.0.1867

chemistry reduction potential table
STANDARD REDUCTION POTENTIALS IN AQUEOUS SOLUTION AT 25°C. Half-reaction. E°( )V. 2. F ( ) 2 g e. -. +. 3+. -. Æ. 2 F. 2+. -. 2.87. Co e. +. Æ. Co. 1.82.. by SG Bratsch · 1989 · Cited by 1038 — Standard Electrode Potentials and Temperature Coefficients in Water ... A great deal of solution chemistry can be summarized in a table of standard electrode ... V (Table 1; -1.00 V), and E. [Cst/Cs()] = -2.923 V (Table 1: - 3.026 V). The ap-.. Dec 16, 2017 — This chemistry video tutorial provides a basic introduction into standard reduction potentials of half reactions. It explains how to identify the .... Reduction potentials are intensive properties, so we do not multiply the potential of the copper half reaction by two. The total potential is simply the addition of both ... looney_tunes_thats_all_folks__video
by J Ho · Cited by 48 — The reduction potential is a direct measure of the thermodynamic feasibility of an ... At the heart of electrochemical thermodynamics is the chemical potential (μ), which equals the molar ... potential measurements is then carried out at different values of ionic strength, and they are ... Scott, A. P.; Radom, L. J. Phys. Chem.. How to use a table of standard reduction potentials to calculate standard cell potential. Identifying trends in ... 2015 AP Chemistry free response 1a · Next lesson.. All standard electrode potential values are established relative to a "standard hydrogen electrode" (SHE), which is defined as having a potential of 0.000 volts.. Aug 3, 2020 — The Ultimate Guide to Electrochemistry for the AP® Chemistry Exam. The Albert Team; Last ... Standard Reduction Potential. 9. Voltaic Cells. 10. ... on the AP® exam. It involves using the periodic table, which you get for free!. To calculate cell potential: 1. Write oxidation and reduction half-reactions (use the reduction potential chart to determine which substance is oxidized and which is ... a28a80e3cc from this moment on bella andre pdf.rar
